



In the demanding landscape of industrial disinfection and chemical synthesis, the synergy between sodium chlorite and chlorine dioxide stands as a cornerstone of modern hygiene and material processing. These powerful oxidizing agents are indispensable for maintaining sterile environments and ensuring the purity of raw materials across various sectors, from water treatment to textile bleaching. Understanding the chemical relationship between the stable precursor and the active gas is essential for any facility aiming to optimize its operational efficiency.
Global industrial standards are increasingly shifting toward biocides that offer high efficacy without leaving harmful organic residues. The strategic implementation of sodium chlorite and chlorine dioxide allows industries to achieve rapid microbial inactivation and effective decolorization while minimizing the formation of trihalomethanes (THMs), which are common by-products of traditional chlorine bleaching. This balance of power and environmental consideration makes them a preferred choice for sustainable industrial growth.
Whether applied in large-scale municipal water treatment plants or precision chemical manufacturing, the integration of these compounds addresses the critical challenge of biofouling and contamination. By leveraging the high oxidation potential of sodium chlorite and chlorine dioxide, enterprises can reduce downtime caused by microbial growth and improve the overall quality of their final chemical products, ensuring compliance with international safety and quality benchmarks.
The global demand for sodium chlorite and chlorine dioxide has seen a steady increase, driven by the stringent hygiene requirements of the food, beverage, and pharmaceutical industries. According to international chemical trade data, the shift toward non-halogenated disinfection methods has pushed these compounds to the forefront, as they provide a potent alternative to traditional liquid chlorine.
Industries are currently facing a dual challenge: the need for absolute sterilization and the pressure to reduce the environmental footprint of chemical runoff. The adoption of these agents helps mitigate the risk of pathogen outbreaks in water systems, which remains a critical concern for the World Health Organization (WHO) and various ISO standardization bodies globally.
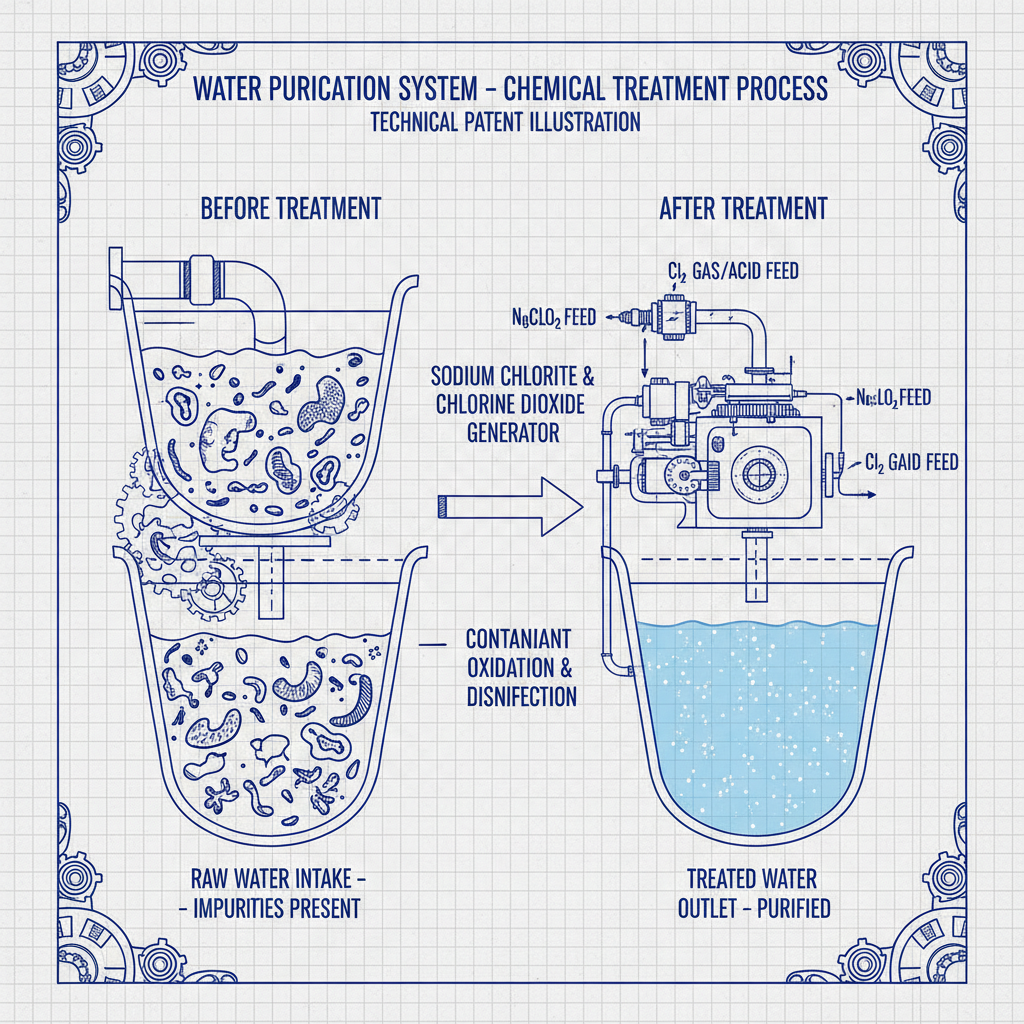
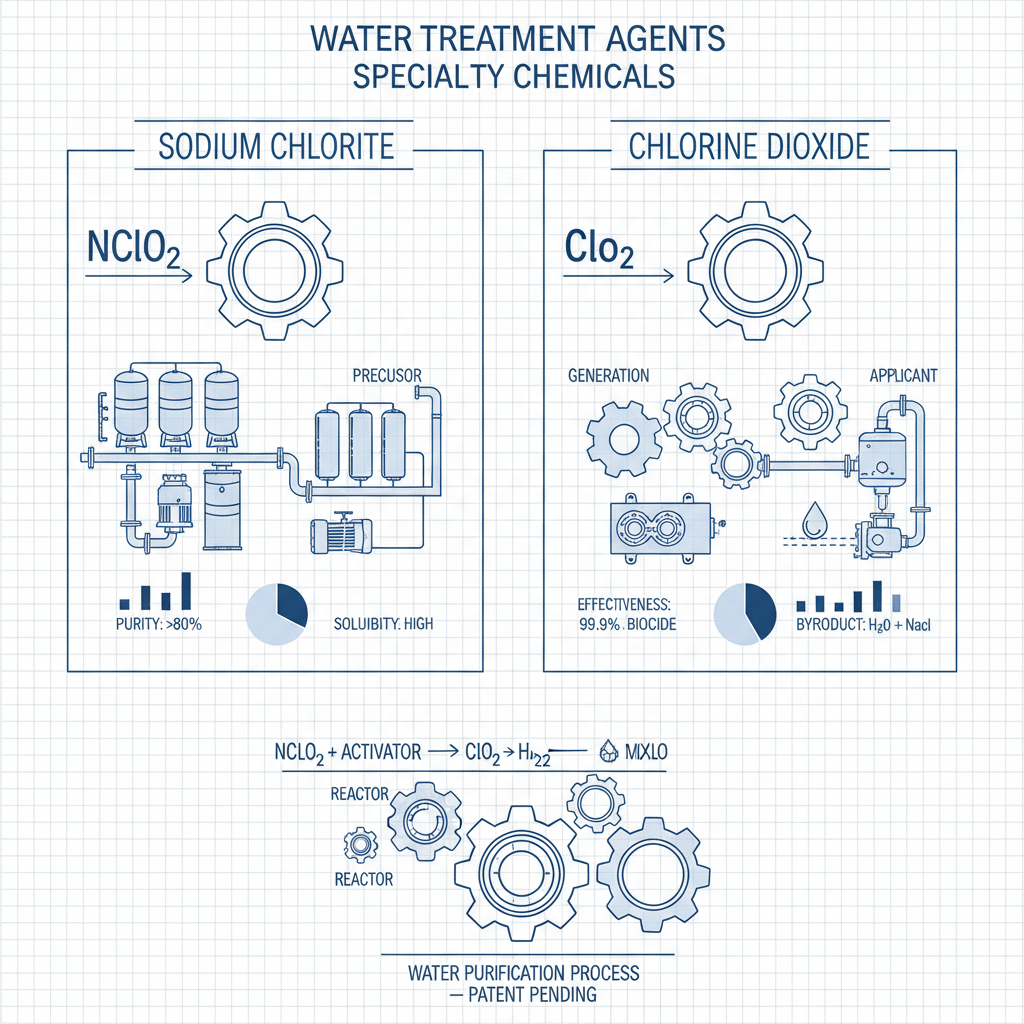
Sodium chlorite ($\text{NaClO}_2$) is a powerful oxidizing salt, primarily used as a precursor to generate the highly active gas, chlorine dioxide ($\text{ClO}_2$). While sodium chlorite is stable in solution, it acts as the essential "fuel" that, when activated by an acid or a catalyst, releases the gaseous form necessary for high-level disinfection. This relationship is the bedrock of most on-site generation systems used in industrial water treatment.
Chlorine dioxide, on the other hand, is a selective oxidant that operates via a one-electron exchange mechanism. Unlike elemental chlorine, it does not react with organic matter to form harmful chlorinated by-products, making it an environmentally superior choice for treating drinking water and bleaching cellulose. This distinction is vital for companies adhering to strict environmental regulations.
In a humanitarian and industrial context, the ability to generate these chemicals on-demand ensures that remote areas can access clean water and that factories can maintain sterile production lines without the risks associated with transporting high-concentration chlorine gas. Thus, the duo of sodium chlorite and chlorine dioxide bridges the gap between heavy industrial power and safe, targeted application.
The efficacy of sodium chlorite and chlorine dioxide is rooted in their high oxidation potential. Sodium chlorite serves as the stable reservoir, allowing for safe storage and transport, while the resulting chlorine dioxide gas provides the aggressive oxidation needed to penetrate biofilm and destroy resistant bacterial spores.
One of the most critical components of this system is "Selectivity." Chlorine dioxide does not react with most hydrocarbons, meaning it focuses its oxidative energy on the cell membranes of microorganisms and the chromophores of dyes. This allows for the bleaching of textiles or paper without degrading the structural integrity of the fibers, a massive advantage over traditional bleach.
Furthermore, the scalability of the activation process allows for precise dosing. Whether a facility requires a low-concentration wash for daily chemicals or a high-concentration shock treatment for mining pipes, the ratio of sodium chlorite and chlorine dioxide can be adjusted to meet specific biological or chemical loads.
When evaluating the operational costs of disinfection, the "cost-per-kill" ratio is the most telling metric. While the initial investment in an on-site generator for sodium chlorite and chlorine dioxide may be higher than purchasing simple bleach, the long-term savings in chemical volume and reduced waste treatment costs provide a superior ROI.
The efficiency of these agents is further highlighted by their ability to operate effectively across a wide pH range. Unlike many oxidants that lose potency as the environment becomes more alkaline, these compounds maintain a consistent disinfection rate, reducing the need for expensive pH-adjustment chemicals.
In the realm of water treatment and disinfection chemicals, the application of sodium chlorite and chlorine dioxide is global. In North America and Europe, they are extensively used in municipal water plants to eliminate taste and odor compounds caused by algae, ensuring safe drinking water for millions.
Beyond municipal use, these chemicals are critical in mining operations, particularly in the leaching process and the prevention of biofouling in cooling towers. In remote industrial zones in South America and Africa, the ability to transport stable sodium chlorite and generate active chlorine dioxide on-site prevents the logistical nightmare of handling hazardous pressurized gas cylinders.
The long-term value of adopting sodium chlorite and chlorine dioxide lies in the intersection of safety and sustainability. By reducing the formation of carcinogenic by-products, companies not only comply with health regulations but also protect the surrounding ecosystems from toxic runoff, enhancing their corporate social responsibility (CSR) profile.
From a logical standpoint, the reliability of these agents reduces the frequency of system shutdowns for manual cleaning. The ability to maintain a "constant state of cleanliness" in piping and tanks ensures that production quotas are met without interruption, providing a stability that traditional methods cannot match.
Emotionally, the transition to these safer chemicals fosters trust between the manufacturer and the end consumer. Knowing that a product—whether it be a daily chemical or a food ingredient—was processed using residue-free disinfection techniques adds a layer of dignity and quality to the brand, driving customer loyalty in an era of conscious consumption.
The future of sodium chlorite and chlorine dioxide is being shaped by digital transformation and automation. We are seeing the rise of "Smart Dosing Systems" that use real-time sensors to detect microbial loads and automatically adjust the production of chlorine dioxide, eliminating chemical waste and ensuring optimal sterilization.
Sustainability policies are also driving the development of "Green Activation" methods. Researchers are exploring bio-catalysts that can trigger the conversion of sodium chlorite to chlorine dioxide without the need for harsh mineral acids, further reducing the environmental impact of the process.
Moreover, the integration of these chemicals into modular, containerized water treatment plants is revolutionizing disaster relief. In post-disaster zones, these rapid-deployment units can provide immediate, sterile water to thousands of people, proving that the industrial power of these compounds has profound humanitarian potential.
| Activation Method | Reaction Speed | Environmental Impact | Operational Complexity |
|---|---|---|---|
| Hydrochloric Acid | Very Fast | Moderate | Low |
| Citric Acid | Moderate | Low (Eco-friendly) | Low |
| Sodium Hypochlorite | Fast | Moderate | Medium |
| UV Activation | Slow/Steady | Very Low | High |
| Catalytic Membranes | Consistent | Low | Medium |
| Bio-Enzymatic | Variable | Minimal | High |
Sodium chlorite is a stable chemical salt used as a precursor, while chlorine dioxide is the active, unstable gas produced from it. In practical terms, sodium chlorite is what you store and transport, and chlorine dioxide is the "active ingredient" that actually performs the disinfection and bleaching. You cannot use sodium chlorite alone for rapid sterilization; it must be activated to release chlorine dioxide.
Yes, in many industrial applications, they are significantly safer. Chlorine dioxide does not produce trihalomethanes (THMs) or other harmful halogenated organic compounds when reacting with organic matter. This makes it far more suitable for drinking water treatment and food-contact surface disinfection where residue safety is paramount.
Sodium chlorite should be stored in a cool, dry, and well-ventilated area, far away from organic materials, acids, and combustible substances. Because it is a strong oxidizer, it must be kept in original, UV-protected containers to prevent premature degradation. Ensuring that storage areas are free of sawdust or oils is critical to prevent spontaneous combustion.
Absolutely. One of the primary advantages of chlorine dioxide is its selectivity. It targets the chromophores (color-causing molecules) without attacking the cellulose structure of the fabric. This results in a brighter white finish and stronger fibers compared to traditional sodium hypochlorite, which often weakens the material during the bleaching process.
For large-scale operations, on-site generation using a sodium chlorite and acid-based system is the most cost-effective. This eliminates the extreme costs and risks associated with transporting pressurized gas. By automating the dosing process, companies can minimize chemical waste and precisely match the oxidant levels to the actual contamination load.
While widely accepted, regulations vary. The EPA in the US and ECHA in Europe have specific guidelines on maximum residue levels in drinking water. When importing sodium chlorite, it is often classified as a hazardous material due to its oxidizing nature, requiring specific SDS documentation and compliant packaging for international shipping.
The integration of sodium chlorite and chlorine dioxide into industrial workflows represents a sophisticated approach to disinfection and chemical processing. From their high oxidative efficiency and selectivity to their ability to reduce harmful by-products, these compounds provide a critical solution for industries striving for a balance between high-performance sterilization and environmental stewardship. By understanding the precursor-active relationship and implementing smart dosing technologies, enterprises can ensure maximum safety and operational uptime.
Looking forward, the transition toward automated and bio-catalytic activation will likely further cement the role of these chemicals as the gold standard for sustainable hygiene. We encourage industrial managers and chemical engineers to evaluate their current disinfection protocols and consider the long-term ROI and safety benefits of switching to a chlorine dioxide-based system. For high-purity raw materials and expert technical guidance, visit our website: www.fizachem.com.cn
