



The chemical industry relies heavily on powerful oxidizing agents to maintain hygiene, ensure water safety, and facilitate complex industrial synthesis. Among these, sodium chlorite and sodium hypochlorite stand out as indispensable tools, each offering unique chemical properties that address a wide array of disinfection and bleaching needs across the globe.
Understanding the nuanced differences between these two compounds is critical for operational efficiency and safety. While both are chlorine-based, their reactivity profiles, stability, and end-products vary significantly, making the strategic selection of sodium chlorite and sodium hypochlorite a deciding factor in the success of water treatment and chemical manufacturing processes.
From the large-scale purification of municipal water supplies to the precision bleaching of textile fibers, the application of these agents ensures that modern standards of sanitation and product quality are met. By exploring the synergy and specific utility of sodium chlorite and sodium hypochlorite, industries can optimize their resource consumption while maximizing antimicrobial efficacy.
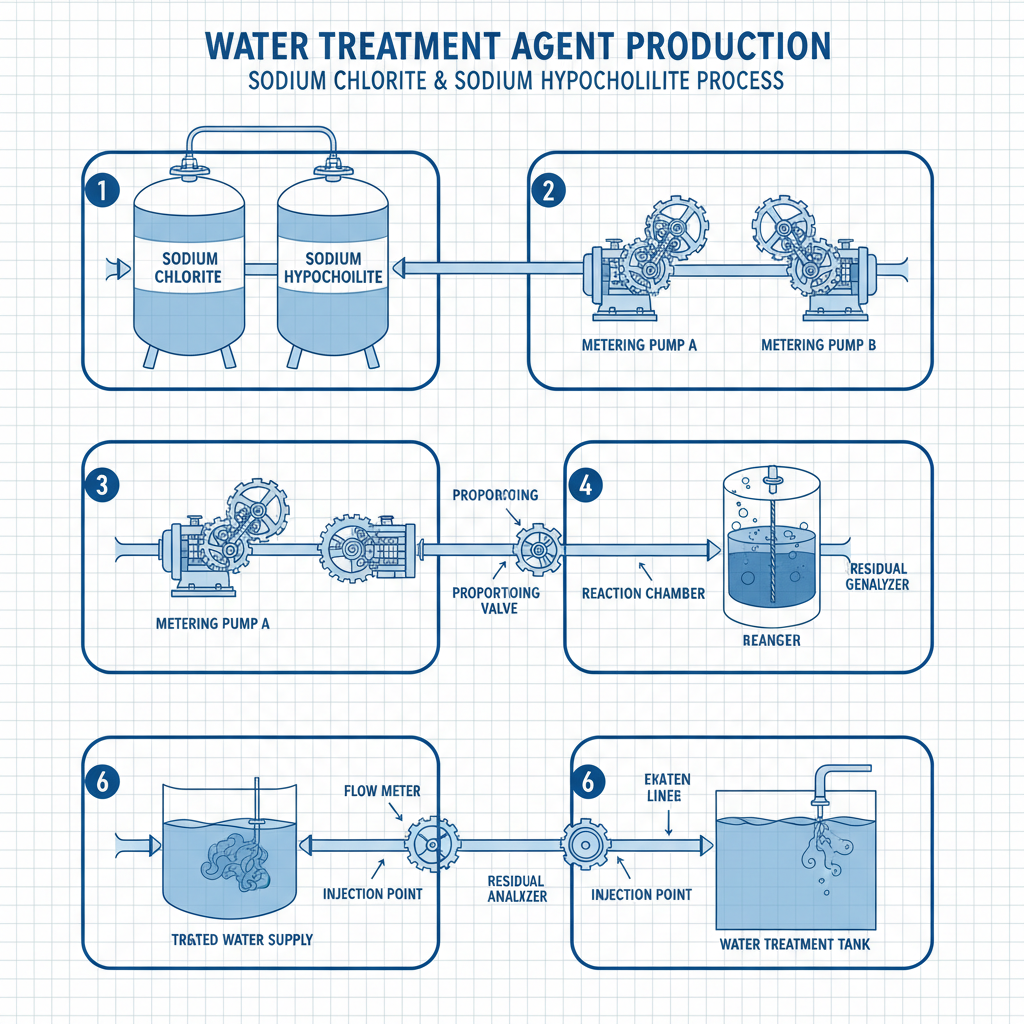
In a world where waterborne diseases and industrial contamination pose constant threats, the global demand for sodium chlorite and sodium hypochlorite has surged. According to standards aligned with ISO and World Health Organization (WHO) guidelines, the ability to rapidly disinfect water and surfaces is a cornerstone of public health, especially in developing regions where infrastructure is limited.
The challenge lies in the balance between efficacy and safety. The volatile nature of these compounds requires precise dosing and handling to avoid environmental toxicity while ensuring that biofilms and pathogens are completely eradicated. This global tension drives the need for high-purity chemicals and sophisticated delivery systems to ensure consistency in performance.
Sodium hypochlorite (NaOCl) is the primary active ingredient in most household bleaches and is widely used for surface disinfection. It works by releasing nascent oxygen and chlorine, which penetrate the cell walls of microorganisms, causing oxidative stress and death. It is the "fast-acting" solution for immediate sterilization needs.
Sodium chlorite (NaClO2), on the other hand, is often used as a precursor to chlorine dioxide (ClO2), a more selective and powerful oxidant. Unlike hypochlorite, chlorine dioxide does not react with organic matter to form harmful trihalomethanes (THMs), making it a preferred choice for high-end water treatment and food processing where residue safety is paramount.
Together, sodium chlorite and sodium hypochlorite represent the spectrum of chlorine-based chemistry. While the former is a direct oxidant, the latter serves as a versatile platform for generating a variety of disinfecting agents, bridging the gap between simple cleaning and advanced chemical sterilization.
The effectiveness of sodium chlorite and sodium hypochlorite depends heavily on concentration and pH stability. In hypochlorite solutions, a high pH is necessary to prevent the release of chlorine gas, ensuring the stability of the solution during transport and storage.
Scalability is another key factor; the ability to produce and deploy these chemicals in bulk—from small-scale daily chemical products to massive water treatment plants—requires a robust supply chain and standardized purity levels to prevent unwanted side reactions.
Cost-efficiency is achieved when the correct agent is matched to the specific contaminant. For instance, using sodium chlorite and sodium hypochlorite interchangeably can lead to waste; hypochlorite is more cost-effective for simple surfaces, while chlorite is superior for deep-cleaning cooling towers.
In the realm of water treatment, sodium chlorite and sodium hypochlorite are utilized to eliminate algae, bacteria, and viruses. In remote industrial zones or post-disaster relief operations, rapid-deployment tablets of these chemicals provide the only line of defense against cholera and other waterborne pathogens.
Beyond water, the paint and fireworks industries utilize these chemicals for specific bleaching and oxidation steps in pigment synthesis. In the agrochemical sector, they help in cleaning equipment to prevent cross-contamination between different pesticide batches, ensuring that crop protection products remain pure and effective.
The long-term value of implementing high-quality sodium chlorite and sodium hypochlorite lies in the drastic reduction of operational downtime. By preventing biofilm buildup in industrial cooling systems, companies save millions in energy costs and equipment replacement.
Moreover, there is an emotional component of trust and safety. When a municipality can guarantee the purity of its water using these agents, it fosters public confidence and societal stability. The innovation in stabilizing these compounds means safer handling for workers, reducing the risk of accidental chemical burns and toxic leaks.
The future of sodium chlorite and sodium hypochlorite is moving toward "Green Chemistry." Researchers are developing on-site generation systems that use electrolysis to produce these agents from simple brine, eliminating the need for hazardous transportation of concentrated chemicals.
Digital transformation is also playing a role. Smart dosing pumps, integrated with IoT sensors, can now monitor water quality in real-time and adjust the injection of sodium chlorite or hypochlorite automatically. This precision prevents over-chlorination and reduces the environmental impact on aquatic ecosystems.
Additionally, we are seeing a shift toward synergistic blends. By combining sodium chlorite and sodium hypochlorite with organic catalysts, the industry is creating "smart oxidants" that only activate in the presence of specific pathogens, further increasing efficiency and reducing waste.
One of the primary limitations of using sodium chlorite and sodium hypochlorite is their tendency to decompose under UV light and heat. This leads to a loss of active chlorine and reduces the efficacy of the treatment process over time.
To overcome this, the industry has adopted opaque, UV-stabilized HDPE packaging and temperature-controlled storage facilities. Expert insights suggest that rotating stock (FIFO) and using stabilizing additives can extend the shelf life of these products by up to 30%, ensuring that the agent remains potent when deployed.
Another challenge is the formation of disinfection by-products (DBPs). The solution involves a phased approach: using hypochlorite for initial bulk disinfection and following up with sodium chlorite-generated chlorine dioxide for precision polishing, which minimizes the creation of harmful organic halides.
| Parameter | Sodium Hypochlorite | Sodium Chlorite | Impact Level (1-10) |
|---|---|---|---|
| Reaction Speed | Very Fast | Moderate (as precursor) | 9 |
| Selectivity | Low (Broad) | High (Targeted) | 8 |
| DBP Formation | Significant (THMs) | Minimal | 10 |
| Cost per Unit | Low | Moderate | 7 |
| Stability | Low (Degrades fast) | High (as salt) | 8 |
| Application Range | General Cleaning | Industrial Purifying | 9 |
The primary difference lies in their chemical action and end-products. Sodium hypochlorite is a direct oxidant used for rapid surface disinfection and bleaching. Sodium chlorite is typically used as a precursor to generate chlorine dioxide, which is a more selective oxidant that does not create harmful trihalomethanes, making it safer for drinking water treatment.
Yes, but with caution. While sodium hypochlorite is effective for quick disinfection, it can be corrosive to certain metals and may lead to the formation of organic by-products. For long-term biofilm control in cooling towers, a sodium chlorite-based chlorine dioxide system is often more efficient and less corrosive.
Both can be used in food-grade applications provided they meet purity standards (like FCC or USP) and are rinsed according to regulatory guidelines. Sodium chlorite (via chlorine dioxide) is often preferred in the food industry because it is highly effective at removing pathogens from produce without leaving a strong chemical taste.
Both chemicals should be stored in a cool, dry, and well-ventilated area, away from direct sunlight and organic materials. Sodium hypochlorite should be kept in UV-resistant HDPE containers to prevent decomposition. Sodium chlorite should be kept away from acids to prevent the premature release of chlorine dioxide gas.
Sodium chlorite is generally considered more eco-friendly for large-scale wastewater treatment when converted to chlorine dioxide. This is because it reduces the formation of halogenated organic compounds, which are persistent environmental pollutants often associated with the use of sodium hypochlorite.
Importing these chemicals requires strict adherence to IMDG (International Maritime Dangerous Goods) codes. You must ensure the supplier provides a comprehensive SDS (Safety Data Sheet), Certificate of Analysis (CoA), and that the packaging is certified for hazardous material transport. Working with a specialized chemical partner like Fizachem simplifies this logistics chain.
In summary, the strategic application of sodium chlorite and sodium hypochlorite provides a comprehensive solution for oxidation and disinfection across diverse industrial sectors. From the rapid sterilization provided by hypochlorite to the precision and environmental safety of chlorite-based systems, these compounds ensure that hygiene standards are maintained and industrial processes remain efficient.
Looking forward, the integration of on-site generation and IoT-driven dosing will further refine the use of these chemicals, minimizing waste and maximizing safety. For companies seeking to optimize their chemical procurement and operational efficacy, investing in high-purity agents is the first step toward sustainable industrial growth. Visit our website: www.fizachem.com.cn
