Sodium chlorite toxicity is a critical concern across diverse industries, from water treatment and disinfection to chemical manufacturing and even specialized applications like paint production. Understanding the potential hazards associated with this compound is paramount for ensuring worker safety, environmental protection, and responsible handling practices. The increasing use of sodium chlorite in various applications necessitates a thorough grasp of its toxicological profile and appropriate mitigation strategies.
Globally, the demand for effective disinfectants and oxidizing agents has risen significantly, leading to increased sodium chlorite production and utilization. This widespread adoption, while beneficial in many respects, also elevates the risk of accidental exposure and potential adverse health effects. Therefore, knowledge about sodium chlorite toxicity is no longer confined to chemists and industrial hygienists; it's a vital piece of information for anyone involved in the supply chain, handling, or potential exposure scenarios.
Addressing sodium chlorite toxicity isn't simply about compliance with safety regulations; it’s about fostering a culture of responsible chemical management and prioritizing human and environmental well-being. The benefits of comprehensive understanding extend beyond preventing immediate harm; they contribute to long-term sustainability and public trust.

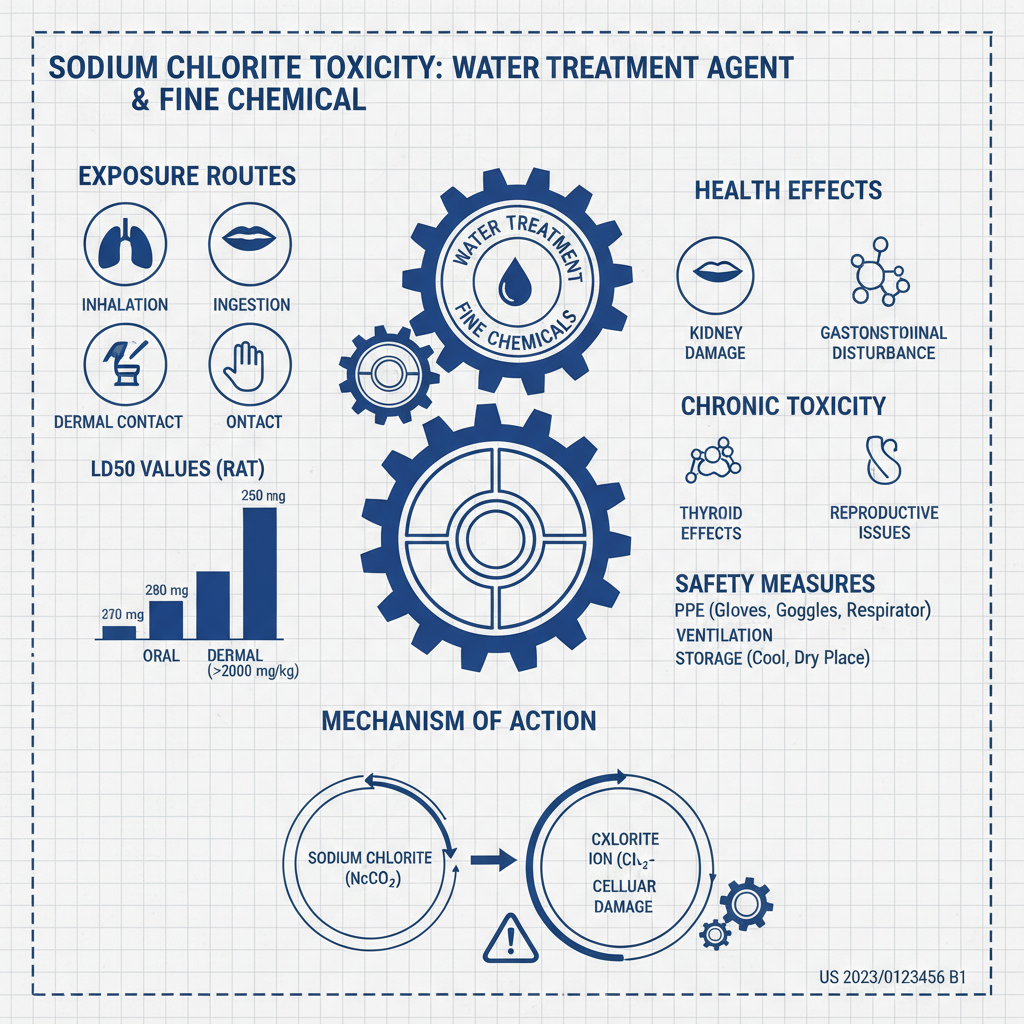
Sodium chlorite toxicity refers to the harmful effects resulting from exposure to sodium chlorite (NaClO2). While used beneficially as a disinfectant and oxidant, its improper handling or excessive exposure can lead to a range of adverse health outcomes. The severity of toxicity depends on the concentration, duration of exposure, and route of entry into the body.
In modern industrial practices, sodium chlorite is frequently employed in various processes, including textile bleaching, pulp and paper production, and water purification. Its effectiveness stems from its ability to release chlorine dioxide, a powerful disinfecting agent. However, this very reactivity is also the source of its potential toxicity, demanding stringent safety protocols and a detailed understanding of its chemical behavior.
The relevance of sodium chlorite toxicity extends far beyond individual workplaces. Globally, increasing demands for potable water and effective disinfection solutions are driving the production and consumption of sodium chlorite. The World Health Organization (WHO) emphasizes the importance of safe water supplies, and sodium chlorite plays a role in achieving this goal in many regions. However, this increased usage also amplifies the potential for accidental release and exposure.
Several industries are heavily reliant on sodium chlorite. The water treatment sector utilizes it for removing contaminants and pathogens. The agrochemical industry employs it in certain formulations, and the pulp and paper sector leverages its bleaching properties. The UN's Sustainable Development Goals, particularly those related to clean water and sanitation, indirectly highlight the need for understanding and mitigating the risks associated with chemicals like sodium chlorite.
A key challenge is the varying levels of safety awareness and enforcement across different countries. Developed nations often have stringent regulations, while developing nations may struggle with implementation and monitoring, increasing the risk of accidental exposure and long-term health consequences.
Sodium chlorite exerts its toxicity primarily through the formation of chlorine dioxide (ClO2) upon contact with acidic environments. This reactive gas is a strong oxidizing agent, capable of damaging cellular components, particularly in the respiratory system and gastrointestinal tract. The mechanism involves the disruption of cellular redox balance and the induction of oxidative stress.
The reaction with gastric acid in the stomach is particularly concerning. Ingestion of sodium chlorite solutions, even in relatively low concentrations, can lead to the release of chlorine dioxide in the digestive system, causing irritation, inflammation, and potentially more severe complications such as methemoglobinemia—a condition where hemoglobin's ability to carry oxygen is impaired.
It’s crucial to understand that the toxicity isn’t solely dependent on the amount of sodium chlorite ingested or inhaled. Individual susceptibility plays a significant role. Individuals with pre-existing respiratory conditions, anemia, or other underlying health problems may be more vulnerable to the adverse effects of sodium chlorite exposure.
Furthermore, prolonged or repeated exposure, even to low levels, can lead to chronic health problems. While acute toxicity often manifests as immediate symptoms, chronic effects may develop over time, making early detection and prevention even more critical.
Exposure to sodium chlorite can occur through several pathways: inhalation, ingestion, skin contact, and eye contact. Inhalation is a significant concern in industrial settings where sodium chlorite is processed or used in gaseous form. Ingestion may result from accidental contamination of food or water, or, unfortunately, in rare cases, intentional misuse.
A comprehensive risk assessment is essential to identify potential hazards and implement appropriate control measures. This assessment should consider the specific processes involving sodium chlorite, the concentration of the solution used, the ventilation systems in place, and the personal protective equipment (PPE) available to workers. Proper labeling of containers and comprehensive training for personnel are also vital components of risk management.
Numerous regulatory bodies worldwide have established standards for the safe handling and use of sodium chlorite. These include organizations like OSHA (Occupational Safety and Health Administration) in the United States, ECHA (European Chemicals Agency) in Europe, and similar agencies in other countries. These regulations typically cover aspects such as permissible exposure limits, labeling requirements, and emergency response procedures.
Compliance with these standards is crucial for minimizing risks and avoiding legal repercussions. It also demonstrates a commitment to worker safety and environmental responsibility, enhancing a company's reputation and fostering trust with stakeholders.
Effective mitigation strategies for sodium chlorite toxicity center around preventing exposure and minimizing the impact of accidental releases. These include using engineering controls such as ventilation systems, implementing administrative controls like safe work procedures, and providing appropriate PPE to workers.
Having a well-defined emergency response plan is also essential. This plan should outline procedures for handling spills, providing first aid, and contacting emergency services. Regular drills and training exercises can ensure that personnel are prepared to respond effectively in the event of an incident.
While acute toxicity is well-documented, the long-term effects of chronic low-level exposure to sodium chlorite are still being investigated. Some studies suggest potential links to respiratory problems, skin sensitization, and other health issues. Further research is needed to fully understand these long-term effects.
Ongoing research is also focused on developing more effective detection methods, improved mitigation strategies, and safer alternative disinfectants. The advancement of nanotechnology and materials science may lead to the creation of novel solutions that minimize the risks associated with chemicals like sodium chlorite.
Understanding the cumulative effects of sodium chlorite exposure, especially in combination with other chemicals, remains a critical area of investigation for toxicologists and environmental health scientists.
| Exposure Duration | Affected Body System | Severity of Effect | Potential Reversibility |
|---|---|---|---|
| Short-term (hours) | Respiratory System | Mild irritation (Rating: 3) | High |
| Medium-term (weeks) | Gastrointestinal Tract | Moderate inflammation (Rating: 5) | Moderate |
| Long-term (months) | Skin | Chronic dermatitis (Rating: 6) | Low |
| Prolonged (years) | Hematological System | Potential for methemoglobinemia (Rating: 7) | Variable |
| Intermittent (variable) | Nervous System | Neurological Symptoms (Rating: 4) | Moderate |
| Combined Exposures | Multiple Organ Systems | Synergistic Toxicity (Rating: 8) | Low |
If someone inhales sodium chlorite fumes, immediately move them to fresh air. Ensure they are breathing and, if not, administer artificial respiration. Seek medical attention promptly, as respiratory irritation and pulmonary edema can develop rapidly. Monitor for signs of shortness of breath or chest pain. Avoid causing the person to vomit and keep them calm and comfortable.
When working with concentrated sodium chlorite solutions, wearing appropriate Personal Protective Equipment (PPE) is crucial. This includes chemical-resistant gloves, safety goggles or a face shield, and a respirator with appropriate filters, especially if there's a risk of airborne exposure. Additionally, a chemical-resistant apron or suit should be worn to protect clothing and skin from splashes.
For sodium chlorite spills, isolate the area and prevent further contamination. Wear appropriate PPE. For small spills, absorb the liquid with an inert material like sand or vermiculite. For larger spills, contain the liquid and contact emergency response personnel. Avoid flushing the spill into drains or waterways and dispose of contaminated materials according to local regulations.
The reversibility of sodium chlorite toxicity depends on the severity and duration of exposure. Mild irritation is usually reversible with symptomatic treatment. However, more severe cases, like methemoglobinemia, require specific medical intervention, such as methylene blue administration, to restore oxygen-carrying capacity. Supportive care, including oxygen therapy, is also vital.
Chronic low-level exposure to sodium chlorite may contribute to respiratory issues, skin sensitization, and potential hematological effects. While definitive long-term studies are still underway, ongoing monitoring and preventative measures are recommended for individuals regularly exposed to the chemical. Maintaining a detailed exposure history is also critical for accurate diagnosis and treatment.
The acidity of a solution significantly impacts sodium chlorite's toxicity. Lower pH levels (more acidic conditions) promote the formation of chlorine dioxide (ClO2), the primary toxic agent. As ClO2 is a gas, it's more readily inhaled and can cause severe respiratory irritation. Therefore, maintaining a neutral or slightly alkaline pH can help reduce the risk of ClO2 formation and toxicity.
Understanding sodium chlorite toxicity is crucial for safeguarding human health and protecting the environment. From its mechanisms of action and diverse exposure pathways to the stringent regulations governing its use, a comprehensive grasp of this chemical's properties is paramount for responsible handling and risk mitigation. By prioritizing safety protocols, investing in ongoing research, and fostering a culture of awareness, we can unlock the benefits of sodium chlorite while minimizing its potential hazards.
Looking ahead, continuous improvement in detection technologies, the development of safer alternatives, and enhanced worker training programs will play vital roles in reducing the incidence of sodium chlorite-related incidents. Ultimately, a proactive and informed approach to chemical management is essential for promoting a sustainable and healthy future. Visit our website: www.fizachem.com.cn to explore our comprehensive range of chemical solutions and safety resources.